This is Part 1 of 4 from The Last Prescription Series.
← The Last Prescription | Part 2 – What is a Cure Worth? →[🧠] Sapien Fusion Deep Dive Series | February 24, 2026
We just found the button
For 30 years, the treatment model for rheumatoid arthritis, lupus, and most autoimmune diseases has rested on a single, uncomfortable truth: we cannot cure them. We can only suppress them. We can quiet the immune system enough that the damage slows. We can keep the fire from spreading. But we cannot put it out.
That truth is no longer true.
The reset is real.
The data is published.
The patients are off their medications.
What goes wrong in autoimmune disease
The immune system is a precision targeting system. Its job is to distinguish self from non-self — to attack pathogens and leave healthy tissue alone. In autoimmune disease, that distinction breaks down. The immune system begins producing autoreactive B cells: cells that treat the body’s own tissue as the enemy.
In rheumatoid arthritis, those cells attack the joints. In lupus, they attack the kidneys, skin, and organs. In myasthenia gravis, they attack the neuromuscular junction, slowly robbing patients of the ability to move, breathe, and swallow. In systemic sclerosis, they attack connective tissue, hardening skin and organs until function fails.
Every biologic therapy ever developed — Humira, Enbrel, Rituxan, Actemra — works by intercepting part of this process. They block the chemical signals that drive inflammation. They deplete some B cells. They slow the damage.
None of them fix the source.
The autoreactive B cells remain. The immune system remains mis-calibrated. Stop the medication, and the disease returns. Which is why 17.9 million RA patients worldwide are on treatment plans with no end date.
What CAR-T actually does
Chimeric Antigen Receptor T-cell therapy was developed for cancer. The principle: take T cells from a patient, engineer them in the laboratory to recognise a specific target on cancer cells, and reinfuse them. The engineered T cells hunt and eliminate everything expressing that target.
In blood cancers, the target is often CD19 — a protein expressed on B cells, both healthy and malignant. CAR-T wipes out the B cell population entirely. In oncology, that is an acceptable trade-off to eliminate leukaemia or lymphoma.
In 2021, rheumatologist Georg Schett at University Hospital Erlangen made an observation that reframed the entire field: autoimmune disease is also driven by B cells
The logic was almost too simple.
If autoreactive B cells are the source of the disease, and CAR-T can eliminate all B cells, then CAR-T should be able to eliminate the autoreactive population and leave the immune system to rebuild from scratch. A new B cell population, reconstituting from stem cell precursors, would emerge naive. Non-autoreactive. Clean.
This is the immune reset. Not suppression. Not management.
Deletion and reconstruction.
Why it works where biologics cannot
The critical distinction is depth of depletion. Biologic therapies like rituximab (an anti-CD20 monoclonal antibody) deplete circulating B cells in the bloodstream, but they leave intact the long-lived plasma cells hiding in bone marrow and lymphoid tissue. Those cells continue producing the autoantibodies that drive damage. The disease persists at a lower simmer.
CAR-T cells are not passive molecules. They are living, proliferating cells. They migrate into tissues. They track down long-lived plasma cells in the niches where monoclonal antibodies cannot reach. They achieve a depth of depletion that no infused drug can match.
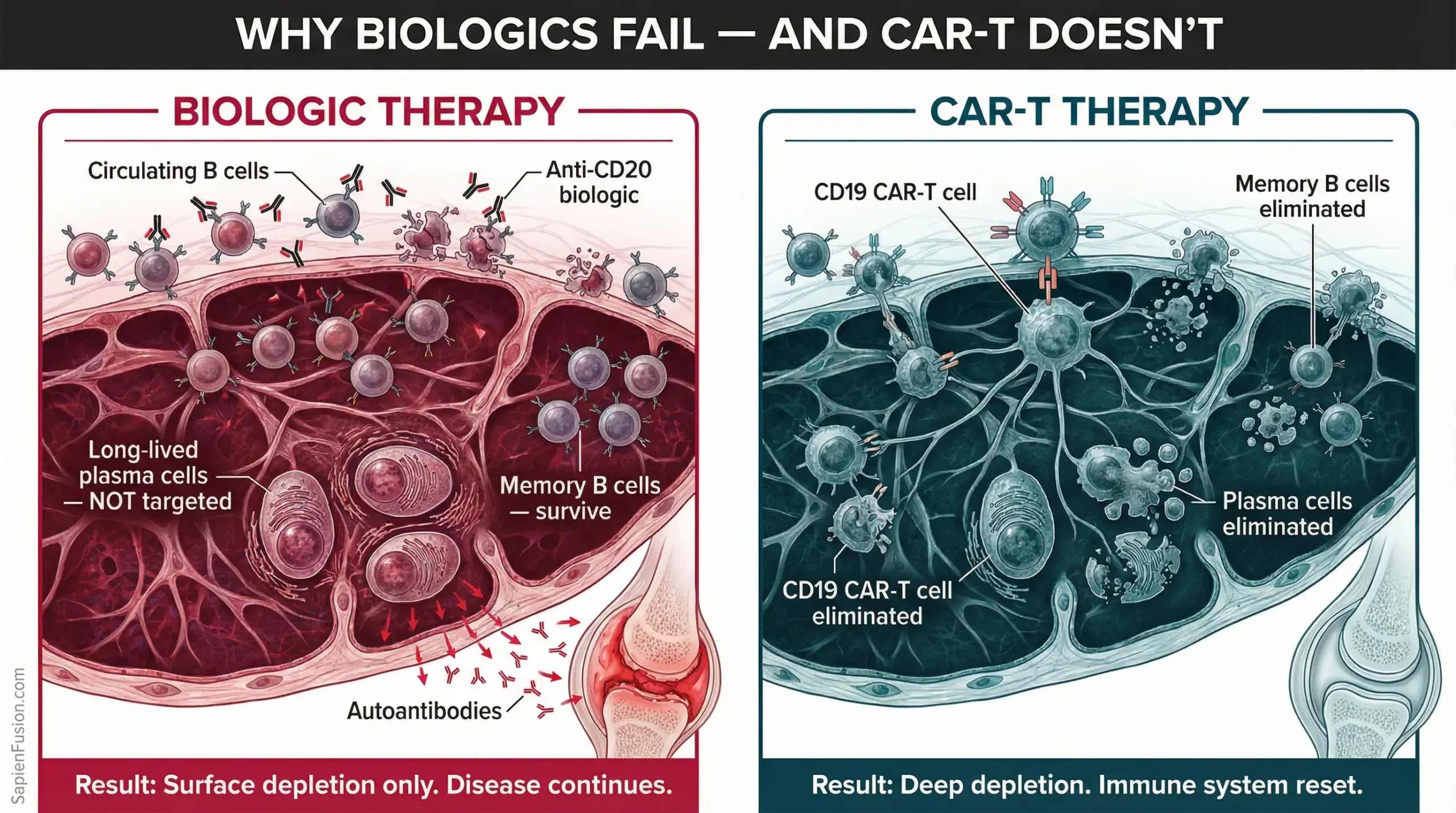
In published SLE case series, following CAR-T treatment, reappearing B cells were predominantly IgM subtype with very low numbers of memory B cells — the signature of a genuinely naive, reconstituted immune compartment, not a temporarily suppressed one. The autoreactive memory had been erased.
In 9 of 10 rheumatoid arthritis patients treated with CAR-T, drug-free remission was achieved. RA-associated autoantibodies converted to seronegative status. The antibodies that had been attacking those patients’ joints for years were gone.
Not reduced. Gone.
The mRNA breakthrough changes who can access this
The original CAR-T manufacturing process is complex, expensive, and requires lymphodepletion chemotherapy — a toxic pre-conditioning regimen that destroys the patient’s existing immune cells to make room for the engineered ones. This requirement confined CAR-T to specialised oncology centres with inpatient facilities and intensive monitoring capacity. For most of the world’s RA patients, it was inaccessible in practice.
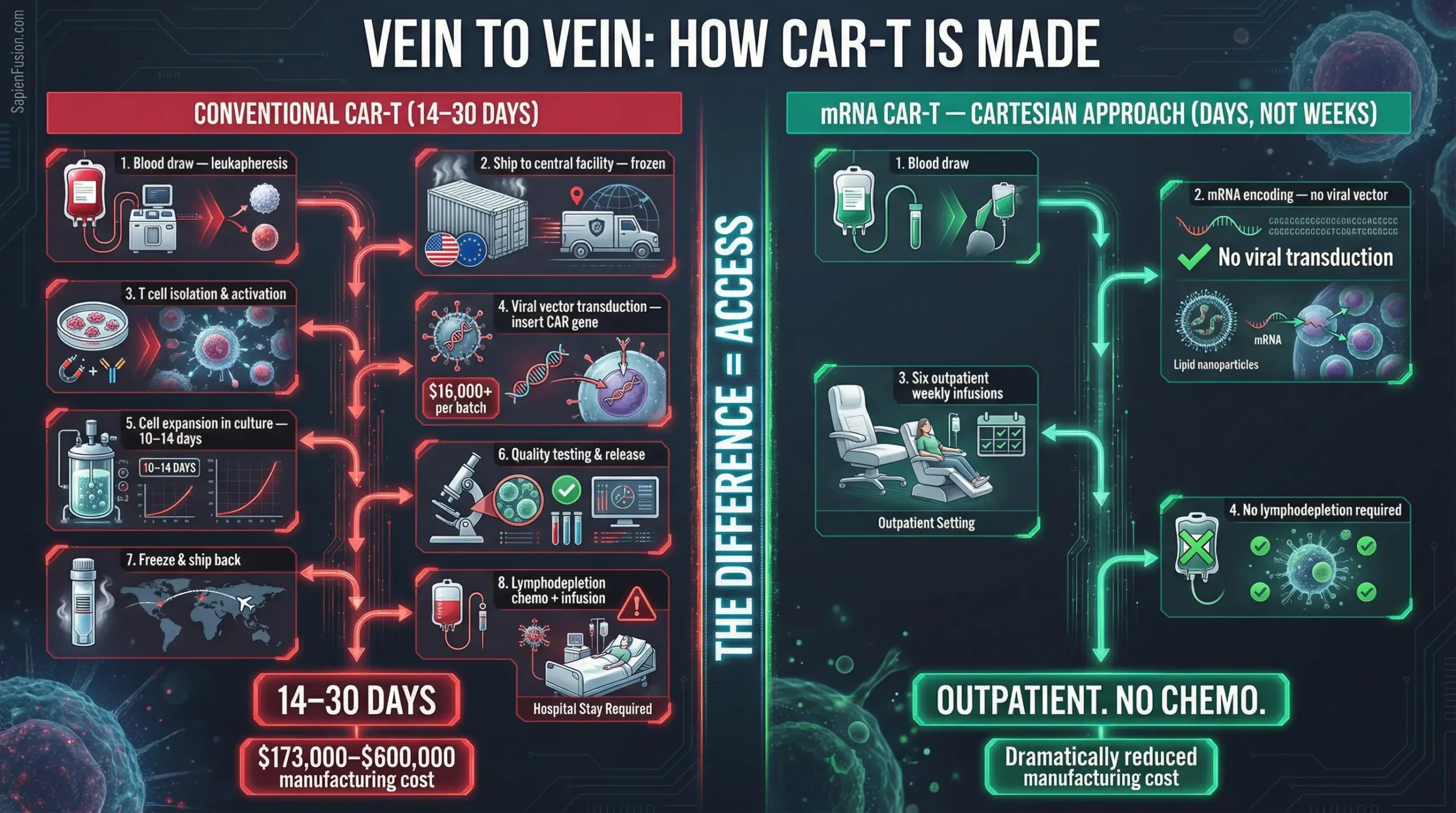
Cartesian Therapeutics, a US-based company, has changed the access equation. Their approach — Descartes-08 — uses messenger RNA rather than permanent viral gene editing to programme the T cells. The CAR construct is encoded in RNA, not integrated into the patient’s genome. The engineered cells function for the therapeutic window, then clear naturally.
This has two consequences that matter enormously for global access.
First: no lymphodepletion required. The chemotherapy pre-conditioning that made traditional CAR-T hospital-dependent is eliminated.
Second: the treatment is administered as six once-weekly outpatient infusions. No inpatient admission. No intensive care backup. The therapy can be delivered in a rheumatology clinic.
In a placebo-controlled Phase 2b trial in myasthenia gravis — a rigorous design, not a case series — Descartes-08 achieved a 66.7% response rate. Zero instances of cytokine release syndrome. Zero instances of neurotoxicity. Patients began tapering their existing medications at month six. Median prednisone dose fell 55% by month twelve.
Outpatient.
No chemotherapy.
Published evidence.
Global access potential.
The reconstitution question
The legitimate scientific question about immune reset is durability. What happens when B cells reconstitute? Do the autoreactive clones return?
Early evidence is cautiously optimistic. In lupus patients with extended follow-up, B cell reconstitution was observed — but the reconstituted population was predominantly naive IgM cells, not memory B cells carrying autoimmune programming. The immune system appears to re-learn tolerance through the normal developmental process: new B cells undergo negative selection against self-antigens before they mature. If the autoreactive clones are not reseeded from surviving precursors, they do not return.
The caveat: long-term follow-up data beyond 12 to 24 months remains limited. The clinical trials are recent. The technology is new. The field does not yet know whether remissions lasting one to two years will extend to five years or ten.
What the field does know: in the patients treated so far, the immune system is not returning to its broken state.
That is not certainty. But it is the strongest signal autoimmune medicine has produced in three decades.
What comes next
The science is established. The mechanism is understood. The clinical evidence is published and peer-reviewed. What remains is scale — the question of how to manufacture this therapy for millions of patients rather than hundreds.
That is the subject of Part 4. Before that, Part 2 examines the economics: what a healthcare system actually saves when a patient stops needing lifetime drugs and returns to productive work. And Part 3 maps the companies racing to bring this to clinic — where they are headquartered, which regulatory agencies they are pursuing, and who stands to win the 17.9 million-patient RA market.
The science has answered the question that rheumatology could not answer for 30 years.
What remains is the execution.
This is Part 1 of 4 from The Last Prescription
All parts in this series:
- Part 1 – Your Immune System has a Factory Reset
- Part 2 – What is a Cure Worth?
- Part 3 – Seven Companies are Racing to be First
- Part 4 – The Cure Works