This is Part 3 of 7 from The Capital Efficiency Paradox Series.
← Part 2: Building the First Longitudinal Women’s Health Dataset | Part 4: $230M to $1B in Four Months→
[🧠] Sapien Fusion Deep Dive | February 16, 2026
Daphne Koller’s Machine Learning-First Pharma
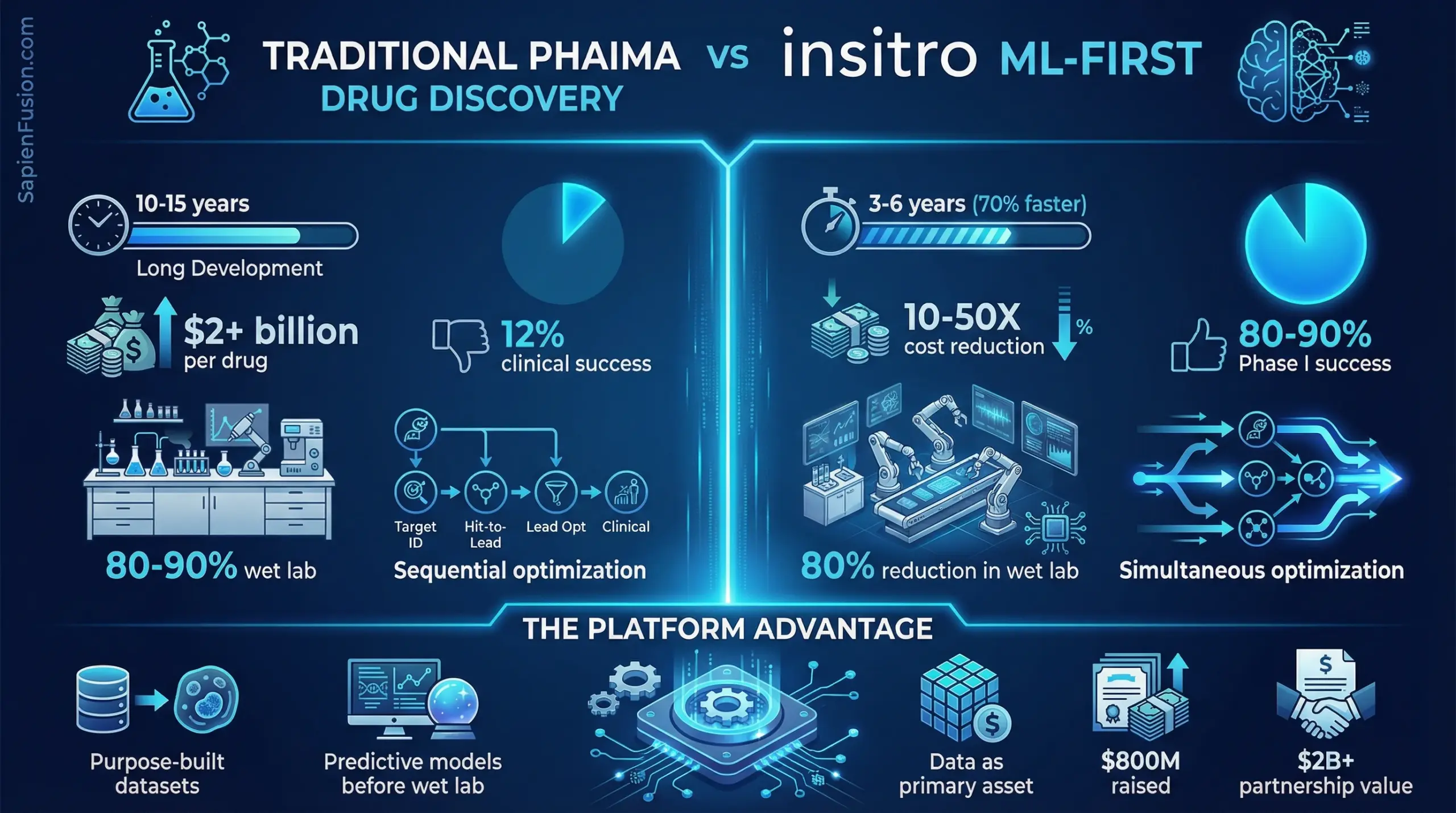
Traditional pharmaceutical R&D takes 10-15 years and burns through $2+ billion per successful drug, with only a 12% clinical success rate. Daphne Koller‘s insitro cuts that timeline to 3-6 years, reduces cost per compound by 10-50x, and achieves 80-90% Phase I success rates.
That’s not incremental improvement through AI tools layered onto existing processes. That’s rebuilding drug discovery from the ground up around machine learning and data. Founded in 2018 with $800M raised to date, insitro has strategic partnerships with Bristol Myers Squibb worth over $2B in potential deal value, multi-program metabolic disease collaborations with Eli Lilly, and validation partnerships with Gilead Sciences. The company expects its first clinic-ready candidates in 2026.
The Intellectual Foundation
Most AI drug discovery companies add machine learning tools to traditional pharma workflows. Koller rebuilt the entire discovery architecture around AI, and it wasn’t speculation. It was grounded in 30+ years pioneering probabilistic graphical models—mathematical frameworks enabling computers to reason about complex systems under uncertainty.
After earning her PhD in Computer Science from Stanford in 1993, Koller spent nearly two decades as the Rajeev Motwani Professor of Computer Science at Stanford, publishing over 300 refereed papers with an h-index exceeding 150. Her 2009 textbook Probabilistic Graphical Models: Principles and Techniques became foundational to the field. The MacArthur Foundation awarded her a “genius grant” in 2004, and she was elected to the National Academy of Sciences in 2023.
The technical expertise wasn’t academic abstraction. Bayes’ theorem scaled to handle millions of potential biological relationships, distinguishing genuine causal effects from statistical noise—exactly the problem drug discovery faces when analyzing high-dimensional cellular data.
The Coursera Detour
In 2012, Koller co-founded Coursera with Andrew Ng, democratizing access to world-class education globally.
She served as co-CEO and then President as the platform reached over 100 million learners. TIME Magazine named her one of the “100 Most Influential People” in 2012, and Fast Company recognized her among “Most Creative People” in 2014. This wasn’t a distraction from computational biology—it was proof that Koller could build platforms serving millions at global scale, not just publish papers.
In 2016, Koller left Coursera to become Chief Computing Officer at Calico, Alphabet’s aging and longevity research company. There she deepened her expertise applying machine learning to biological datasets, genomic information, and disease modeling. By 2018, the thesis crystallized: the pharmaceutical industry was underutilizing the AI revolution. Traditional drug discovery remained labor-intensive, slow, and expensive—ripe for disruption through systematic application of machine learning.
The Platform Thesis
Traditional pharma treats data as a byproduct of drug development. Clinical trials generate data that helps understand whether a drug worked, but only after billions have been spent. Koller inverted the paradigm: generate large-scale, purpose-built biological datasets explicitly designed to fuel machine learning models, then use predictive models to dramatically accelerate and derisk discovery.
The core problem traditional pharma creates is sequential optimization. Companies optimize for binding affinity first, then check metabolic stability, then brain penetration, then toxicity, then manufacturability. The result: molecules highly potent in lab assays fail in clinical trials due to inadequate pharmacokinetics, toxicity, or inability to reach disease tissue.
insitro’s approach performs simultaneous optimization across the complete pharmacological profile from initial iteration. The platform solves for binding affinity AND selectivity AND metabolic stability AND brain penetration AND safety all at once, treating pharmacology as a design constraint rather than an afterthought.
The Founding Advantage
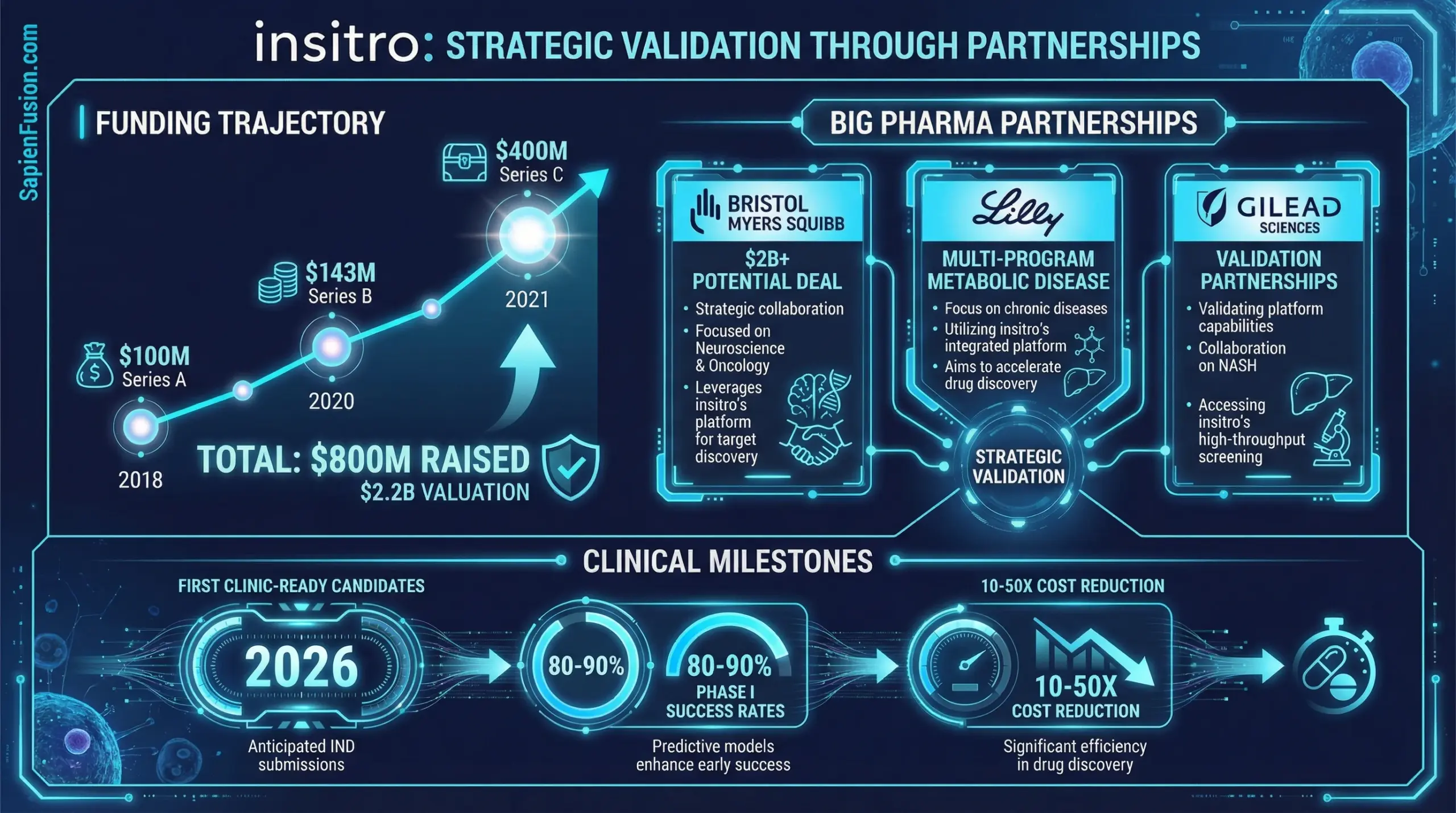
Koller founded insitro in 2018 with a $100M Series A from an elite syndicate: ARCH Venture Partners, Foresite Capital, Andreessen Horowitz, Google Ventures, and Third Rock Ventures. That’s deep biotech plus AI expertise signaling conviction before the company had generated a single data point.
The funding trajectory continued aggressively. A $143M Series B followed in 2020 led by Andreessen Horowitz, then a massive $400M Series C in 2021 led by Canada Pension Plan Investment Board. Combined with approximately $150M from non-dilutive pharma partnerships, insitro has raised around $800M total with a post-Series C valuation estimated at $2.2B.
Here’s the capital efficiency insight: the large initial raises enabled building integrated platform infrastructure—automated labs, imaging systems, compute clusters—that generates multiprogram value. Each improvement to ML models benefits all programs simultaneously, not just single candidates. Traditional biotech companies make separate infrastructure investments per program. insitro built a single platform infrastructure amortized across unlimited programs.
Technical Platform Architecture
POSH Platform: Pooled Optical Screening in Human Cells
Published in Nature Communications in December 2025, the POSH platform validated a breakthrough innovation integrating pooled CRISPR screening with high-content imaging and self-supervised machine learning. Historically, researchers faced a brutal trade-off. Traditional CRISPR screening could interrogate thousands of genes but provided limited readouts, typically just a single reporter protein—high throughput but low phenotypic depth. High-content imaging captured rich multi-dimensional phenotypes but was logistically prohibitive at genome scale—low throughput but high phenotypic depth.
POSH synthesized both approaches. The platform combines pooled CRISPR screening with Cell Painting, a multi-component fluorescent staining technique, plus Vision Transformer deep learning models trained on raw pixel images. The results speak for themselves: self-supervised models discovered biologically meaningful gene networks directly from morphology, recovering 2.5x more functional gene relationships compared to conventional analyses. The platform identified novel regulators of mTORC1 signaling, including AURKAIP1 and HSD17B10, validated through orthogonal experiments. Across 1,640 genes screened, POSH reconstructed known biological networks like the proteasome and Golgi-ER without any prior hypotheses.
Capital efficiency emerges from eliminating the historical compromise. A single integrated platform simultaneously achieves scale AND phenotypic depth.
ChemML Platform: End-to-End Small Molecule Design
Built through internal development plus the acquisition of Haystack Sciences, ChemML integrates multiple capabilities into a unified engine. Quantitative Adaptive Libraries provide hundreds of millions of binding affinity and selectivity data points. ADMET prediction models were trained on 40 years of Eli Lilly drug chemistry data through strategic collaboration. An iterative design-make-test engine becomes smarter each cycle by combining computational predictions with experimental data. The platform runs on 192 H100 GPUs providing computational power for high-end ML and physics-based simulation.
The strategic differentiation is architectural. Traditional medicinal chemistry optimizes potency first and hopes ancillary properties align. ChemML optimizes binding affinity, selectivity, metabolic stability, brain penetration, and safety profile simultaneously from cycle one. This architectural difference explains why AI-native companies achieve 80-90% Phase I success rates compared to 40-65% for traditionally discovered drugs.
TherML Platform: Modality-Agnostic Therapeutic Design
In January 2026, insitro acquired CombinAbleAI, an Israeli AI therapeutics company from AION Labs, launching the TherML platform. CombinAbleAI brought physics-informed ML models for antibody and complex biologic design, pre-trained on over 100,000 molecular dynamics simulations.
TherML’s integration created full modality coverage: small molecules through ChemML, antibodies through CombinAbleAI, oligonucleotides through insitro’s internal capabilities, plus multi-specific proteins. The capital efficiency advantage is stark. Traditional approaches require building separate discovery engines for small molecules, antibodies, and oligonucleotides. insitro’s unified platform selects therapeutic modality based on biology rather than chemistry constraints, with investment compounding across modalities.
The Partnership Model
insitro doesn’t license single candidates to pharma. Instead, it provides platform access for multi-target discovery within therapeutic areas while retaining core datasets and ML models.
The Bristol Myers Squibb collaboration, announced in 2020, focuses on ALS and frontotemporal dementia. The deal structure included $50M upfront, $20M in near-term milestones, over $2B in potential discovery, development, regulatory, and commercial milestones, plus royalties on net product sales. In October 2025, insitro and BMS extended the partnership, applying ChemML to a novel ALS target discovered in Phase 1. The total potential deal value now exceeds $2B, translating from biology discovery during 2020-2025 to molecule design during 2025-2026 within a single partnership.
Stephen Hitchcock, a scientific advisor with over 30 years in pharma R&D, explained the significance: “ChemML’s approach of simultaneously optimizing across the complete pharmacological profile represents a fundamentally different approach enabling scientists to target indications that would otherwise remain intractable, particularly CNS disorders.”
The Eli Lilly collaboration came in two waves. In September 2025, insitro trained ML models on 40 years of Lilly’s drug chemistry data. The resulting models integrated into ChemML, while Lilly developed TuneLab, a platform providing biotech access to their data. In December 2024, three strategic agreements focused on metabolic diseases, particularly MASLD. insitro secured options to in-license Lilly’s clinical-stage GalNAc delivery technology for two siRNA programs and collaborative antibody therapeutic development for a third metabolic target. insitro retains full global rights while Lilly receives milestones and royalties.
The inaugural partnership with Gilead Sciences in April 2019 validated the platform approach. Focused on NASH and MASLD, the deal included $15M upfront, $35M in near-term milestones, and $200M potential per program across up to five programs. Gilead provided clinical trial data while insitro applied its platform for target identification.
The partnership structure consistently follows the same logic: pharma funds research, insitro retains platform ownership and core datasets, and both parties share downstream commercial upside for specific targets within defined therapeutic areas.
The Capital Efficiency Mathematics
The economics tell the transformation story. Traditional pharmaceutical R&D requires 10-15 years from concept to approval, costs over $2B per successful drug including failed programs, and achieves just 12% clinical success for drugs entering trials. Large permanent facilities and teams must be maintained regardless of program outcomes, creating massive fixed costs.
AI-native drug discovery through insitro’s model compresses timelines to 3-6 years, with some programs reaching clinical candidates in just 18 months. Cost per compound drops 10-50x compared to traditional pharma. Phase I success rates hit 80-90% versus 40-65% for traditionally discovered drugs. Computational infrastructure scales with programs while the core platform amortizes across unlimited programs, converting fixed costs to variable costs.
The headcount differential is equally striking. Most pharma companies employ 50,000-70,000 people. insitro runs with approximately 230-350 employees following a 2024 optimization, achieving comparable throughput with 1/200th the workforce. The difference isn’t automation replacing humans—it’s rebuilding the discovery architecture so ML models handle optimization that traditionally required armies of medicinal chemists conducting sequential trial-and-error.
Most pharma companies layer AI tools onto existing workflows. insitro rebuilt from the ground up around ML and data, positioning the company as infrastructure rather than just another drug developer.
Why This Represents Capital Efficiency
Five interconnected advantages compound over time. First, platform leverage across unlimited programs means core infrastructure investments (automated biology, compute clusters, ML frameworks) benefit every program simultaneously. One improvement to POSH helps every disease target, unlike traditional approaches where lab investments benefit single programs.
Second, multimodal coverage through unified platform architecture provides strategic flexibility. insitro selects optimal therapeutic modality based on target biology rather than organizational chemistry constraints, with learnings from small molecule programs informing antibody design and vice versa.
Third, partnership model derisk allows pharma to fund research while insitro retains platform ownership and datasets. Each partnership generates new training data that improves the platform for all subsequent programs.
Fourth, frontloading platform infrastructure investment creates compounding returns. Traditional pharma amortizes infrastructure costs across single program lifecycles. insitro amortizes across every program forever, with marginal cost per program approaching zero as platform matures.
Fifth, validated first-mover infrastructure advantage means while competitors build platforms, insitro already demonstrated clinical candidate generation and established pharma partnerships, creating a widening moat as the dataset grows.
Pattern Recognition Across Career
The capital efficiency pattern repeats across Koller’s career arc. As a Stanford professor during 1995-2005, she pioneered probabilistic graphical models, creating mathematical infrastructure enabling countless AI applications. Through Coursera in 2012, she built online platforms democratizing education, reaching 100+ million learners globally. With insitro beginning in 2018, she’s deploying ML-first drug discovery architecture achieving 10-50x cost reduction with 2-3x faster timelines.
Each transition represents seeing which domains are structurally ready for ML-driven transformation, building the platform infrastructure that enables it, then capturing disproportionate value as the platform scales. From MacArthur “genius grant” recipient to Coursera co-founder to founder of a $2.2B+ valued AI pharma company reshaping drug discovery—that’s capital efficiency as recognizing which architectural transitions matter.
Current Position and Outlook
Koller currently serves as Founder and CEO of insitro, leading the company toward 2026 clinical milestones that will validate or refute the fundamental thesis: machine learning, when applied systematically to purpose-built biological datasets, can dramatically reshape pharmaceutical R&D economics.
The bleeding edge reality: if first clinical candidates succeed, insitro validates a new drug discovery architecture. If they fail or underperform, the company demonstrates that pharmaceutical biology remains too complex for current ML approaches despite massive capital investment. Either outcome provides signal about AI’s true capabilities versus limitations in biological systems.
From probabilistic reasoning to 100 million Coursera learners to rebuilding drug discovery—that’s capital efficiency as identifying which domains are structurally ready for ML-driven transformation.
Learn More About Daphne Koller
Professional Profiles:
Current Position:
- Founder and CEO at insitro
Recognition:
- National Academy of Sciences (2023)
- National Academy of Engineering (2011)
- MacArthur Fellowship (2004)
- TIME 100 Most Influential People (2012)
- TIME “100 Most Influential People in AI” (2024)
Company Resources:
- insitro Platform – AI-driven drug discovery technology
- insitro Pipeline – Therapeutic programs
- Coursera Instructor Page – Educational contributions
Media Coverage:
This is Part 1 of 7 from The Capital Efficiency Paradox
All parts in this series:
- Part 1: Capital Efficiency Through Humanoid Telepresence
- Part 2: Building the First Longitudinal Women’s Health Dataset
- Part 3: $800M to Rebuild Drug Discovery
- Part 4: $230M to $1B in Four Months
- Part 5: From 20 Researchers to 2.7 Billion Downloads
- Part 6: Capital Efficiency Through Government-to-Commercial Path
- Part 7: The Capital Efficiency Playbook
- Research Methodology and Source Verification for the Capitol Efficiency Paradox Series

